Written by Kristen Kusek, Former Communications Director for USF CMS

Tim Conway
Sure, Tim Conway, Associate Professor of chemical oceanography, appreciates it when a dolphin, shark or tarpon surfaces by the seawall outside his office window in St. Petersburg, Florida. But he’s the kind of scientist who gets even more excited by the things you can’t see in the ocean – things like the trace metal iron, which he’s been studying since 2006.
Conway’s long-time collaborator, Dr. Jessica Fitzsimmons, an associate professor at Texas A&M University, recently invited him to join her in writing an invited comprehensive review about iron in the Annual Review of Marine Science. The review takes you on a deep dive into marine iron biogeochemistry and iron isotopes. We sat down with Dr. Conway to get more of a helicopter view of the bigger-picture questions.
Q: Please start us out with a short primer on iron in the ocean.
A: Iron is important because it’s influential as a lever on the carbon cycle. It’s essential for everything that lives in the ocean and has a global impact on where phytoplankton grow and how they take up carbon – but it really doesn’t want to be in seawater. It’s insoluble in seawater.
The iron that is in the ocean comes from atmospheric dust, sediments, or hydrothermal venting. However, iron is tricky to study mostly because there’s precious little of it – as in, one-billionth of a gram of iron (one nanogram) in every liter (1000 grams) of seawater. That’s a ratio akin to one small fraction of one person out of the entire global population.
In addition, we have to use clean techniques to study iron because everything we use to collect it is a potential source of contamination. Ships are rusty, equipment is full of metals, and in the early days it was hard to measure iron accurately. Even as recently as the 1950s, measurements of iron were three orders of magnitude higher than those taken today – all because the proper clean techniques were not being used or available.
Q: What are the top take-away messages of the review?
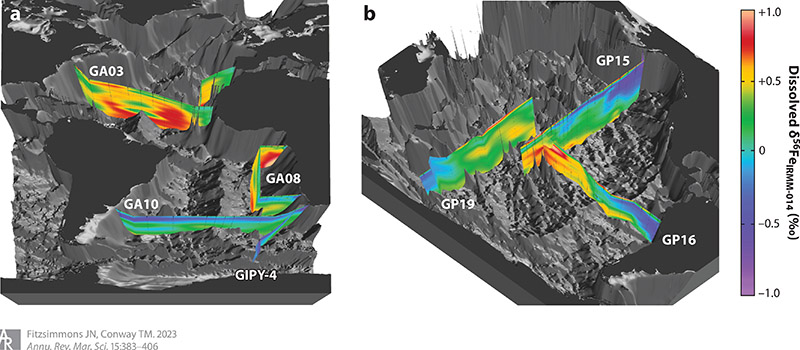
A: The top take-away is that in 2012 we had about 50 datapoints for iron isotopes in the entire ocean. Now, we have roughly 3000. So no matter how you interpret the data, we have a huge amount of it available, which is exciting. The other key point is how useful iron isotopes have been in driving forward understanding. In the review we cover four case studies that demonstrate what we’ve learned – from developing a new way to understand how sediments dissolve to improving our ability to tease out whether iron in atmospheric dust was coming from pollution or natural sources.
Q: How have iron isotopes helped in the study of iron in the ocean?
A: The understanding of iron isotopes is a relatively new development in the field. We only started working with them in the late 2000’s – roughly 2007 to 2009 – but they have been incredibly transformative to the field. Every piece of iron has a specific isotope ratio that is like a chemical fingerprint. That fingerprint can change depending on the sample’s chemistry. By measuring the isotopes, we can learn about the history of the sample and where the iron came from. So if you just measure iron itself, you have no idea where it came from – but you can tell if you measure iron isotopes. The most common of the four iron isotopes is 56 (56Fe); in fact, about 95% of all the iron in the ocean is Iron-56. The other just 5% comprises all the other isotopes, making measuring isotope ratios even more challenging than just iron itself!
Q. What are some of the potentially most transformative avenues of research related to iron?
A. One of the largest challenges that we’ve had since the beginning is how we measure dissolved iron – the portion that goes through a filter. It’s really hard to know what this iron really is. For example, is the iron bound to humics or siderophores (small compounds that help microorganisms accumulate iron)? Is it truly dissolved as ions? Is it a soup of ions, particles, and organics? Likely all of the above, and scientists are still developing a range of cool techniques to figure this out. One aspect that my lab is focused on, in partnership with Oregon State University, is using iron isotopes to look at specific parts of the dissolved iron pool – not the whole thing. If it’s one specific compound that is grabbing the iron, what’s its chemical fingerprint? This is tough to do but we’re trying.
Q. What has been the most successful aspect of the GEOTRACES program?
A. GEOTRACES has produced these huge datasets from 35 different countries that can be collated and that all utilize the same methodology. Before GEOTRACES, people did their own thing, their own way – but now we have a data collection “cookbook,” for example, which is incredibly helpful. We also have an international standards committee, which I serve on currently, and we evaluate how well various science teams are using the methods. It’s amazing and helpful that we’re all working together. From a US perspective, so many things are now measured on the same cruise using multiple tracers. In fact, most of the periodic table is being measured on every GEOTRACES cruise in some way.
Q . Is there a segment of the ocean that is critical to further explore, or explore further, in order to better our understanding of iron in the ocean?
A. Yes, and hopefully No. Here’s what I mean. In its early phases the GEOTRACES program focused a lot on the Atlantic Ocean and gradually moved into the Pacific. Until 2018 there was very little data from the North Pacific, and there is still very little data from the South Pacific and around Antarctica. This isn’t surprising because these are harder areas to study but they are so critical to understand. The good news: the US GEOTRACES program literally just finished a cruise aboard the R/V Roger Revelle in the South Pacific that my student, Dylan Halbeisen, participated on (Dec 2022 – Jan 2023). A second US GEOTRACES cruise will visit the Amundsen Sea in late 2023. The South Pacific, the Antarctic Circumpolar Current and the Antarctic region are the areas we still need to focus on because the Antarctic sits there as this hub of ocean circulation. The things that happen in Antarctica – upwelling in particular – don’t stay in Antarctica and control the distribution of trace elements across all the other oceans.
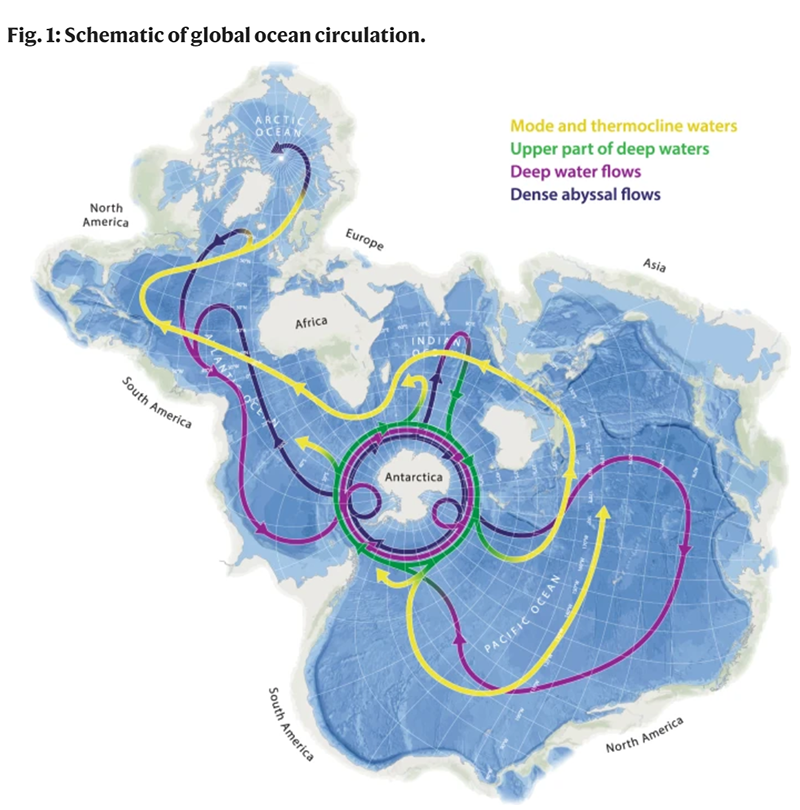
The centrality of the Southern Ocean to the planetary-scale circulation is clear when viewed on a Spilhaus projection. It is the most prominent site globally where deep waters (purple) rise to the surface, exchange heat and carbon with the atmosphere, and then sink back to the ocean interior. The densest waters formed (black) can store carbon in the abyss for centuries or longer. This contrasts with injection into shallower layers (yellow), from which the carbon can be returned to the surface on much shorter timescales. Credit: Nature Communications (Nat Commun) ISSN 2041-1723 (online).
Q. If you could wave a magic wand to better understand iron, science or science fiction, what would you do?
A. The biggest challenge we have with iron is as soon as you put it in a bottle, you change what’s going on. Iron loves to stick to things. The chemistry of iron is impacted by the container. So I’d like to somehow suspend the seawater and analyze it without the need for a container, by making use of a Star Trek type force field or something. That would be a huge advancement for our work!
Q. Take a step, even a leap, back, and help us understand what you see as the top, big picture questions facing chemical oceanographers today.
A. Tough question! I'm going to have to focus in a little and talk about the trace elements...I think we still have a lot to learn, with a lot of our understanding of the trace elements coming in the last ten years. I think one of the biggest challenges today is still understanding the form of elements in seawater - whether they are present as truly dissolved ions, complexed to poorly known organic molecules, or even present as colloidal particles. The second one is really getting to grips with the relationship between trace elements and 'biology'...Hopefully a successor to GEOTRACES will be a project called 'biogeoscapes', which will aim to provide synergy and look in much greater detail about the role trace metals play for marine DNA, proteins and microbes.
Need more iron? Check out this recent textbook chapter Conway co-authored that focuses on the sampling and analyticl techniques needed to measure iron and other trace metals (manganese, copper, nickel, zinc) in the oceans.